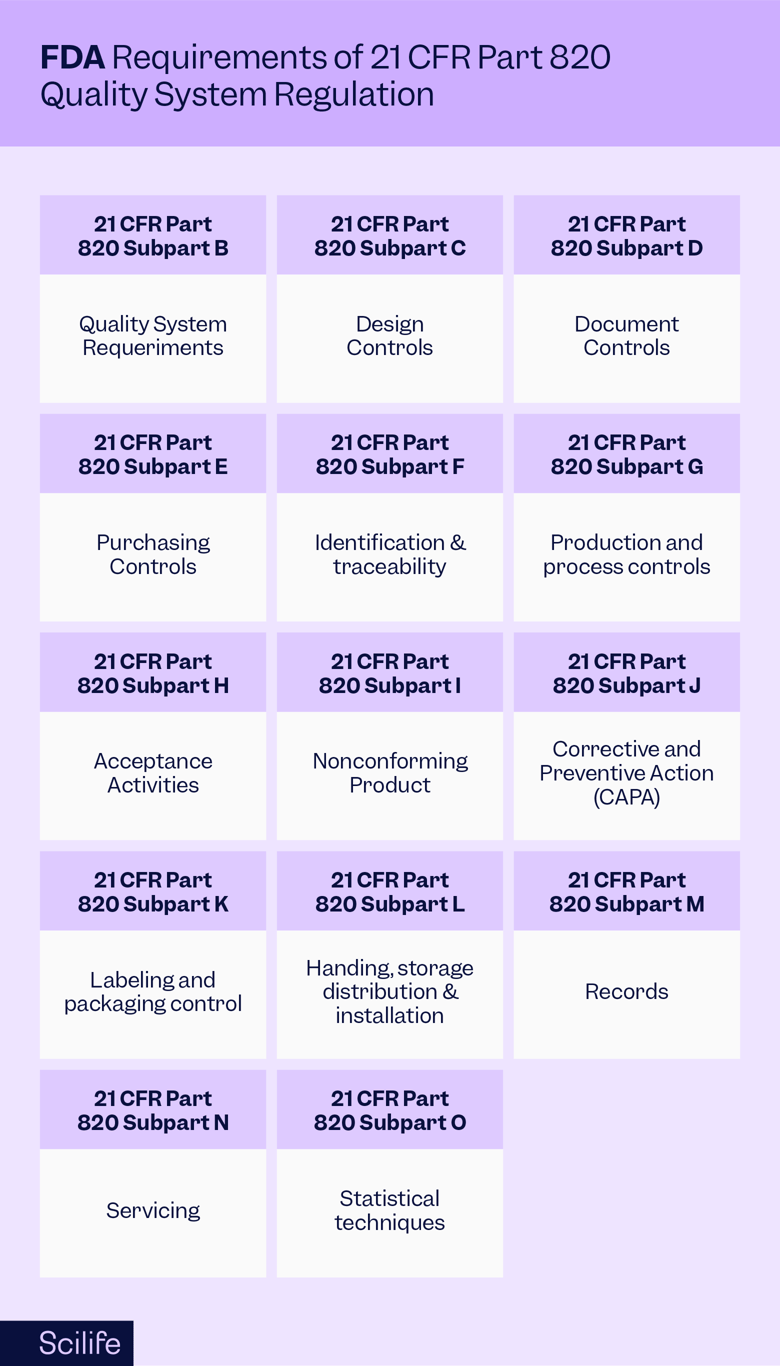
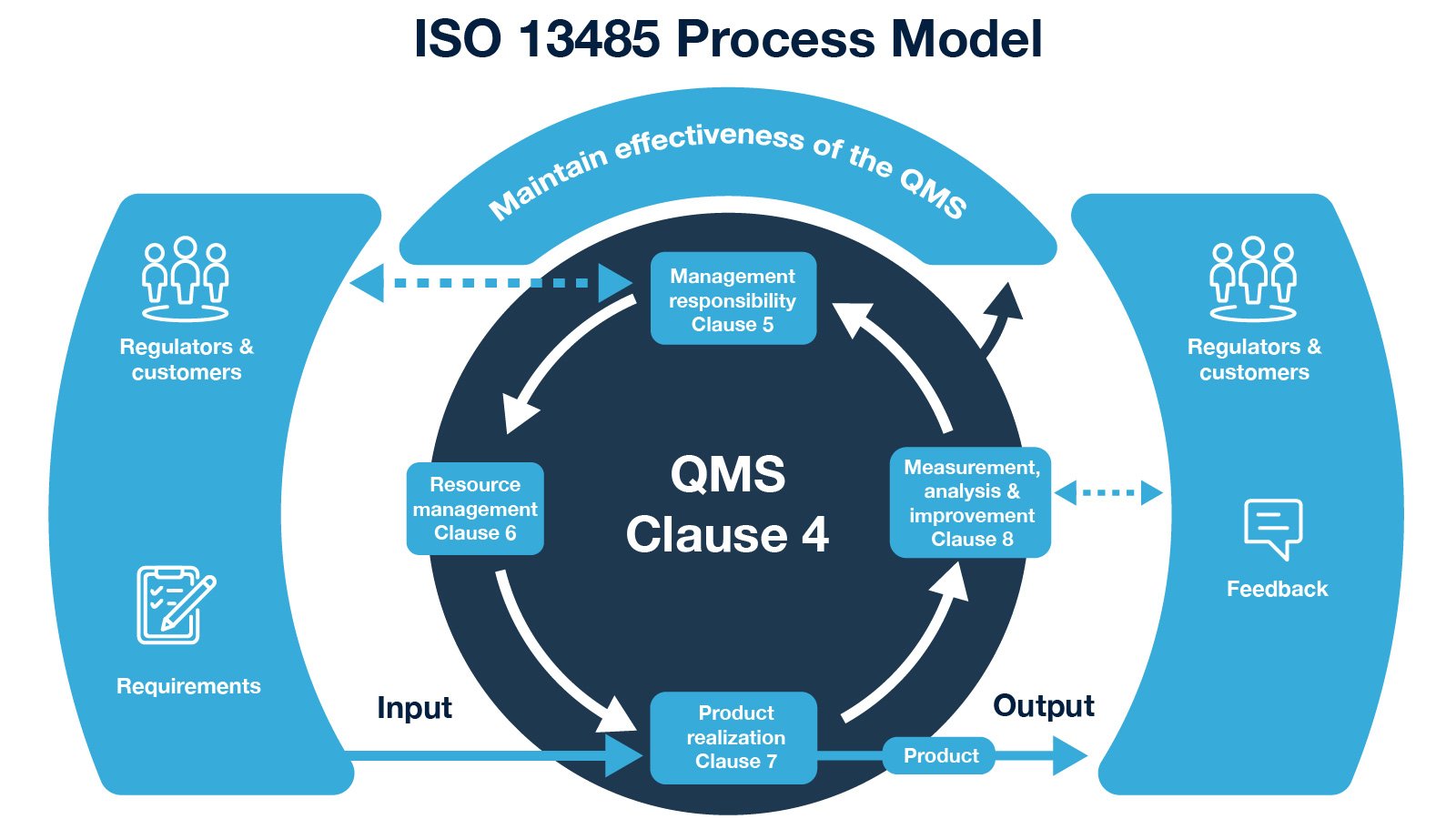
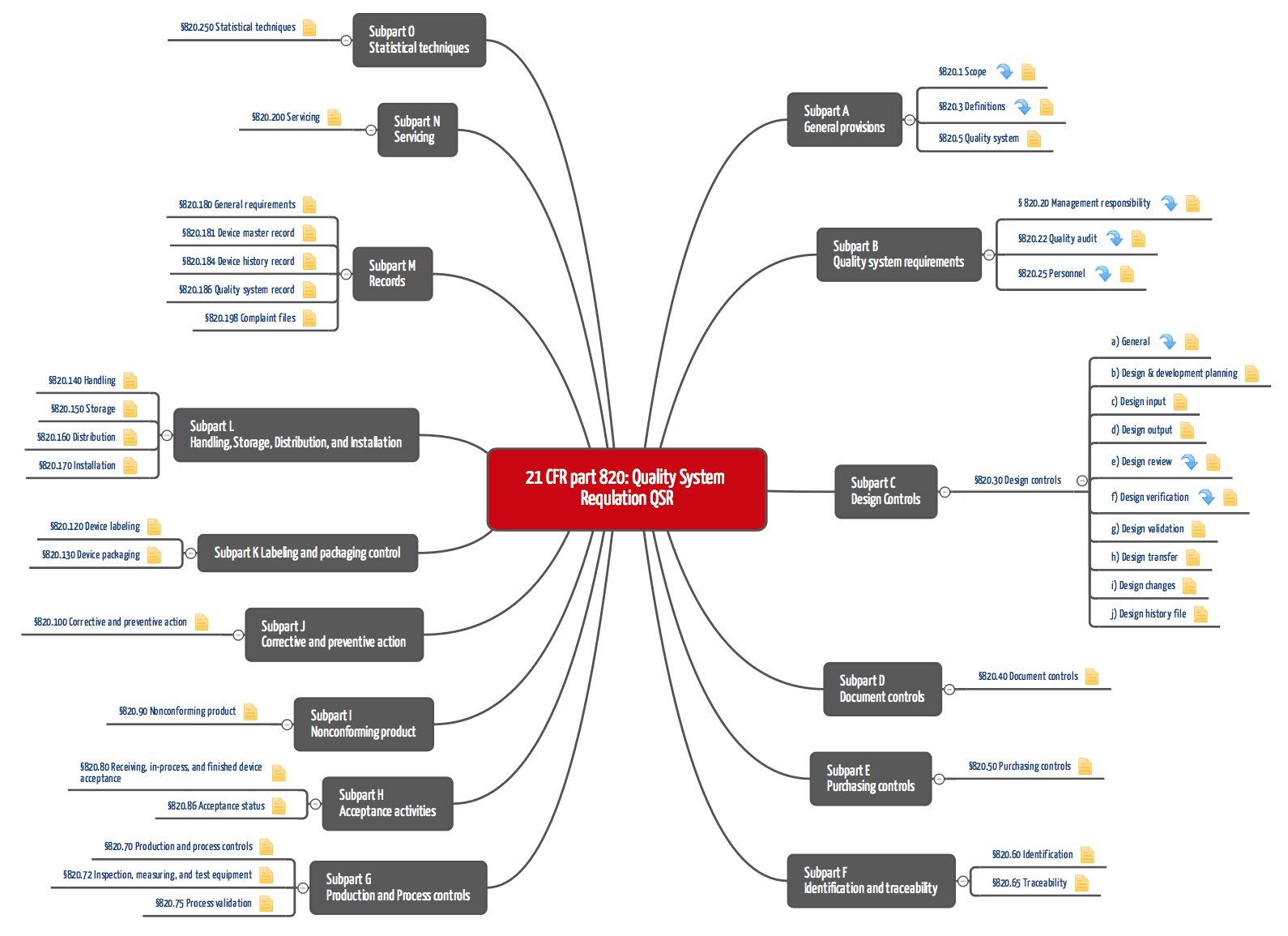
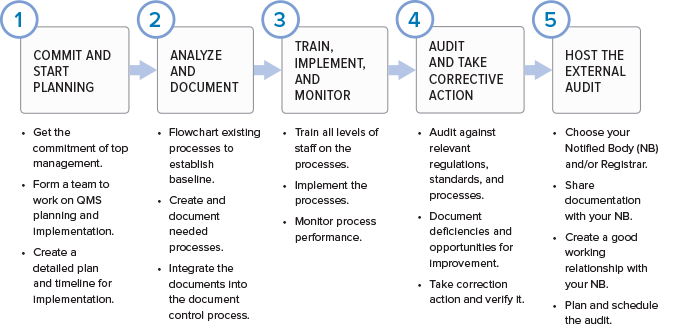
The FDA and Worldwide Quality System Requirements Guidebook for Medical Devices, Second Edition | ASQ

21 CFR Part 820 - Quality System Regulation | 21 CFR 820.30 Medical Device Design Control Guidelines - YouTube

FDA Quality System Regulation for Medical Devices (21 CFR Part 820): A Practitioner's Guide to Management Controls (sections 820.20 Management ... 820.22 Quality Audit, and 820.25 Personnel) : Daugherty, Mr D G: Amazon.fr: Livres